Future of the Mycoplasma Testing Market: Automation, Accuracy, and High-Throughput Solutions
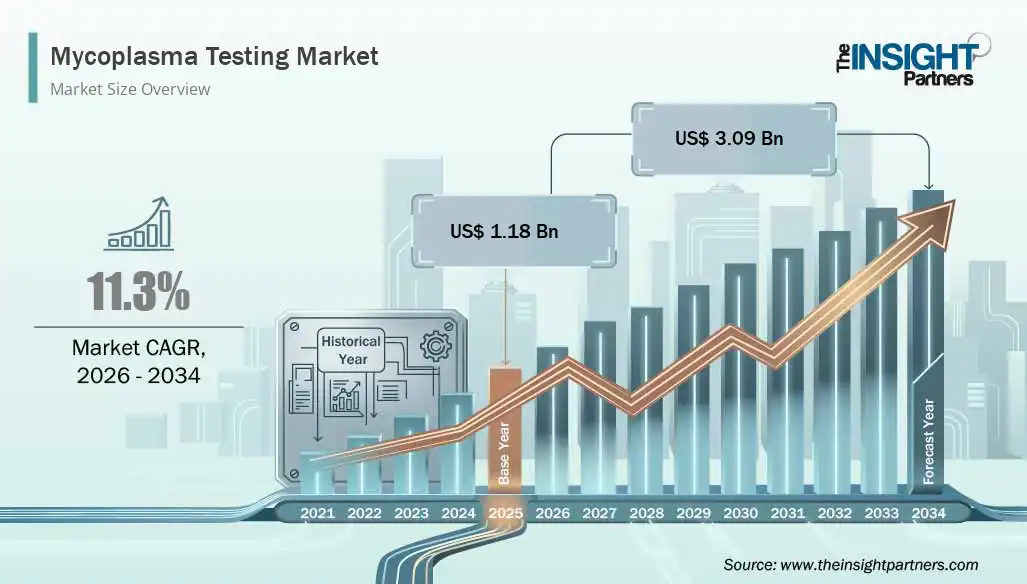
The global Mycoplasma Testing Market is experiencing strong growth driven by rising biopharmaceutical production, increasing contamination risks in cell culture, and expanding regulatory scrutiny. Advancements in rapid PCR-based detection technologies are reshaping the Mycoplasma Testing Market. According to industry analysis, the market is projected to grow from US$ 1.18 billion in 2025 to US$ 3.09 billion by 2034, registering a CAGR of 11.30% during 2026–2034. Mycoplasma contamination continues to be one of the most significant quality and safety concerns in biologics manufacturing, making testing an essential step in pharmaceutical, biotechnology, and research workflows.
Mycoplasma are small bacteria lacking cell walls, making them difficult to detect and resistant to many antibiotics. Their presence can compromise cell cultures, vaccines, gene therapies, and biologics, leading to product loss, regulatory penalties, and patient safety risks. As a result, demand for advanced mycoplasma testing technologies is increasing rapidly across pharmaceutical companies, contract research organizations (CROs), academic labs, and biomanufacturing facilities.
Download Sample PDF Copy:
https://www.theinsightpartners.com/sample/TIPRE00003516
Market Overview
Mycoplasma testing refers to methods used to detect mycoplasma contamination in cell cultures, raw materials, biologics, vaccines, and advanced therapies. Testing is mandatory in biopharmaceutical manufacturing due to global regulatory requirements. Technologies used include PCR, ELISA, direct assay methods, microbial culture, and nucleic acid amplification tests (NAT).
The rising complexity of biologics manufacturing and the expansion of cell and gene therapies are transforming quality control practices. This transformation is creating substantial opportunities for testing kit manufacturers, reagent providers, and service providers.
Key Market Drivers
Rapid Growth of Biopharmaceutical Manufacturing
One of the strongest drivers of the mycoplasma testing market is the global expansion of biologics production. Biopharmaceutical products such as monoclonal antibodies, vaccines, recombinant proteins, and cell therapies rely heavily on mammalian cell culture systems, which are highly susceptible to mycoplasma contamination. The global shift toward biologics is increasing testing frequency at multiple stages of production, including raw material screening, cell bank testing, in-process monitoring, and final product release testing. The rise in biologics pipelines and biosimilar development has significantly increased the need for routine contamination monitoring.
Increasing Adoption of Cell and Gene Therapy
The rapid expansion of cell and gene therapy pipelines is a major growth catalyst. These therapies use living cells and viral vectors, making contamination risks extremely high. Strict regulatory requirements, high production costs, and the need for rapid batch release testing are pushing manufacturers to invest in faster and more accurate mycoplasma testing solutions.
Stringent Regulatory Guidelines and Compliance
Global regulatory agencies have implemented strict guidelines requiring mycoplasma testing in biologics manufacturing. Compliance includes mandatory testing of master and working cell banks, viral vaccines, and biologics before product release. Non-compliance can lead to recalls, production shutdowns, and reputational damage, encouraging pharmaceutical companies to adopt advanced testing platforms.
Rising Incidence of Contamination in Cell Culture
Mycoplasma contamination remains a widespread issue in research and manufacturing laboratories. Contamination often occurs through personnel, raw materials, cross-contamination, or inadequate sterilization practices. Because mycoplasma infections are difficult to detect visually, routine screening is becoming a standard laboratory practice.
Growing Outsourcing to CROs and CMOs
Pharmaceutical and biotech companies increasingly outsource research and manufacturing to CROs and CMOs to reduce costs and accelerate product development. These service providers must comply with strict quality standards, driving demand for high-throughput and automated mycoplasma testing solutions.
Technological Advancements in Rapid Detection Methods
Traditional culture methods can take weeks, but modern PCR-based and nucleic acid amplification technologies can deliver results within hours. Automation, real-time PCR, and high-throughput screening are significantly improving efficiency and enabling faster product release timelines.
Expansion of Vaccine Production Worldwide
Global vaccine production has increased significantly, requiring rigorous contamination testing to ensure safety and efficacy. The expansion of vaccine pipelines for infectious diseases and emerging pathogens continues to drive testing demand.
Increasing Investment in Life Sciences Research
Academic institutions, biotech startups, and research labs are expanding work in stem cells, cancer research, immunology, and molecular biology. Routine contamination monitoring has become a standard practice, boosting demand for testing kits and reagents.
Market Segmentation Overview
By Product: Kits and reagents, instruments, and services. Kits and reagents dominate due to recurring demand.
By Technique: PCR, microbial culture, ELISA, direct assay, and others. PCR-based testing is expected to grow fastest.
By End User: Pharmaceutical and biotechnology companies, CROs, and academic research institutes.
Regional Insights
North America leads due to strong pharmaceutical infrastructure and regulatory compliance. Europe maintains a significant share driven by biosimilar production and research investment. Asia-Pacific is expected to grow at the fastest pace due to expanding biopharmaceutical manufacturing and outsourcing activities.
Competitive Landscape – Top Players
Leading companies operating in the market include Thermo Fisher Scientific, Merck KGaA, Lonza Group, Charles River Laboratories, Bio-Rad Laboratories, Roche Diagnostics, Agilent Technologies, Sartorius AG, Eurofins Scientific, and PromoCell GmbH. These companies focus on innovation, automation, and rapid testing technologies to strengthen their market positions.
Future Market Opportunities
The market is expected to expand further due to personalized medicine growth, biosimilar pipeline expansion, automated testing adoption, rapid microbial testing innovations, and increasing global healthcare investments.
Future Outlook
The global Mycoplasma Testing Market is poised for strong growth through 2034. Rising biologics production, increasing regulatory requirements, and the expansion of advanced therapies are driving sustained demand for advanced testing solutions. Continuous technological innovation and growing outsourcing trends will further accelerate market expansion.
About Us
The Insight Partners is a one stop industry research provider of actionable intelligence. We help our clients in getting solutions to their research requirements through our syndicated and consulting research services. We specialize in Semiconductor and Electronics, Aerospace and Defense, Automotive and Transportation, Biotechnology, Healthcare IT, Manufacturing and Construction, Medical Device, Technology, Media and Telecommunications, and Chemicals and Materials.
Contact Us
Ankit Mathur | The Insight Partners
E-mail: ankit.mathur@theinsightpartners.com
Phone: +1-646-491-9876
Also Available in : Korean | German | Japanese | French | Chinese | Italian | Spanish
- Art
- Causes
- Crafts
- Dance
- Drinks
- Film
- Fitness
- Food
- Juegos
- Gardening
- Health
- Home
- Literature
- Music
- Networking
- Other
- Party
- Religion
- Shopping
- Sports
- Theater
- Wellness
